Description
DESCRIPTION
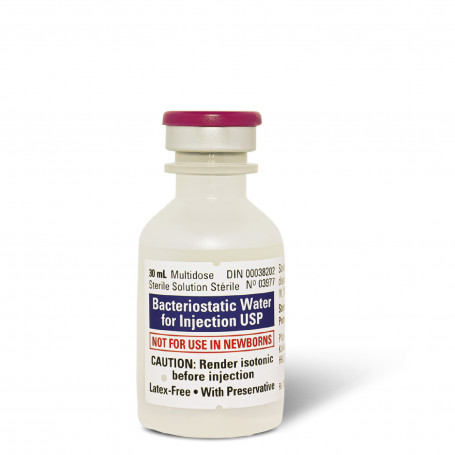
Bacteriostatic water for injection is sterile, non-pyrogenic water that contains 0.9% benzyl alcohol (9mg/ml). The alcohol is added to the water as a bacteriostatic preservative.
When prepared in this way (a mixture of sterile water and benzyl alcohol), bacteriostatic water allows for multiple withdrawals from a single 30ml plastic vial within the 28 days following its first use. The addition of the benzyl alcohol suppresses or stops the growth of bacteria that could potentially contaminate humans.
Once a vial of bacteriostatic water is open, it must be used within the next 28 days. After 28 days the preservatives added to the water no longer maintain its quality and the vial should be disposed of.